MitoCatch delivers healthy mitochondria to diseased cells
In many diseases, dysfunctional mitochondria play a central role. These small structures supply cells with energy. Researchers have now developed the system MitoCatch, which delivers healthy donor mitochondria specifically to the cells that need them most urgently.
15 April 2026
Mitochondrial dysfunction is at the root of many currently untreatable diseases. These include neurodegenerative diseases such as Parkinson’s, degeneration of the optic nerve, and certain forms of heart failure. While researchers have experimented with transplanting healthy mitochondria into damaged cells, traditional methods are often inefficient. They also cannot guarantee that the new mitochondria actually reach the places where they are most urgently needed.
In the journal Nature, researchers led by Prof. Dr. Botond Roska at the Institute of Molecular and Clinical Ophthalmology Basel (IOB) and the University of Basel have reported on a new technology called MitoCatch. This technology enables mitochondria from healthy donors to be introduced specifically into cells that are most severely affected by disease.
Match-maker for mitochondria and cells
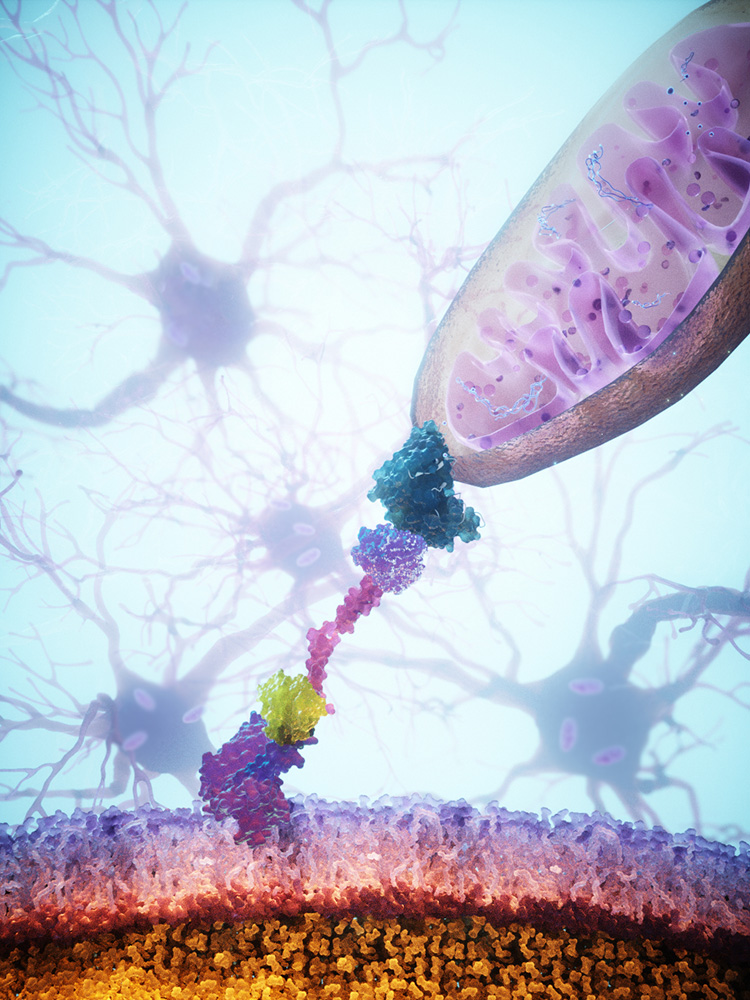
The MitoCatch system is based on engineered docking proteins that guide mitochondria specifically into the right cells. Researchers at IOB and the University of Basel have developed three different strategies for this. In MitoCatch-C, the target cells are equipped with docking proteins on the cell surface so that the new mitochondria can attach to them and be taken up into the cell. However, this requires modifying the target cells outside the body and then returning them to the organism.
With the MitoCatch-M approach, this is not necessary. In this case, the donor mitochondria are modified instead. They are given docking proteins that guide them to the right target cells and allow them to attach only there. In the third variant, MitoCatch-Bi, both the target cells and the donor mitochondria remain unchanged. Instead, a bispecific docking protein serves as a bridge that connects the mitochondria to the cell surface. By specifically adjusting these docking proteins, researchers can control how efficiently and selectively mitochondria reach different cell types.
Temurkhan Ayupov and Dr. Verónica Moreno-Juan, co-first authors, and their colleagues tested the system in various cell types from both mice and humans. It showed that MitoCatch reliably delivers mitochondria to neurons as well as retinal, heart, endothelial and immune cells. Once inside the cell, the mitochondria remain functional. They move, fuse and divide, which is crucial for the normal energy management of a cell.
An approach for damaged optic nerves and retina
The research team also showed, using tissue samples from deceased patients, that donor mitochondria improve the survival of neurons from people with diseases of the optic nerve, while also promoting the regeneration of retinal ganglion cells after injury. So far, this method is well tolerated in animal models, with no detectable immune reaction.
MitoCatch opens up an entirely new approach to precision mitochondrial medicine. The technology overcomes a long-standing obstacle to delivering donor mitochondria specifically into diseased cells and could therefore revolutionise the treatment of mitochondrial diseases.
This article is based on a press release by the IOB.
Original publication
Temurkhan Ayupov, Verónica Moreno-Juan et al.
Cell-type-targeted mitochondrial transplantation rescues cell degeneration
Nature (2026), doi: 10.1038/s41586-026-10391-0