Local immune coordination in the lung reveals a new layer of defense
When a virus enters the lungs, the immune system has to react fast. The lung maintains its own community of immune cells capable of mounting a local defense on the spot. Researchers from the University of Basel now describe the role of a specialized group of cells that orchestrates this local response, directing neighboring immune cells to work together. Their findings could pave the way for new inhalable vaccines against respiratory viruses such as influenza.
24 March 2026 | Martina Konantz
Vaccines usually train the immune system through the blood, but respiratory infections like influenza start in the airways. “If you want to stop a virus, you have to catch it at the door,” says Professor Carolyn King. Her research team at the Department of Biomedicine of the University of Basel studied mice infected with influenza.
In the journal Immunity, the scientists now report a previously unappreciated subtype of helper T cells, immune cells that normally reside in lymph nodes but migrate to the lung during infection. Once there, these T cells begin producing a protein called HIF-1α, typically known as a cellular stress sensor but which can also be switched on by immune activation signals.
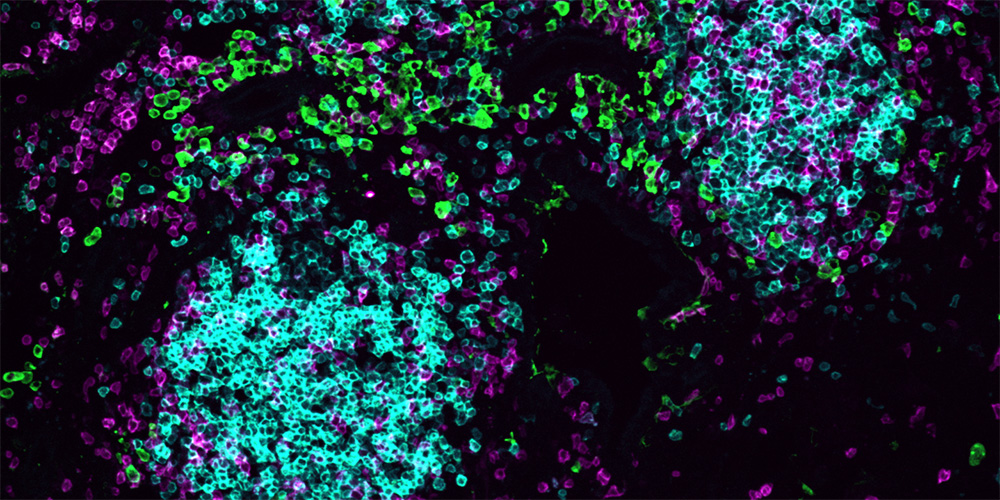
Mapping the lung’s immune architecture
The group used advanced imaging methods to map where different immune cells are positioned in the lung. During infection, the lung develops small, temporary immune hubs – structures that function like miniature lymph nodes, bringing immune cells together in one place to organize a response. They found that the T cells producing HIF-1α are strategically located at the outer edges of these hubs where they release a signaling molecule called interleukin-21 (IL-21). This messenger protein activates neighboring immune cells such as macrophages, antibody-producing B cells, and natural killer cells, allowing them to mount a coordinated local defense.
To better understand the role of HIF-1α, the researchers used a mouse model that enabled them to switch off this molecule at a precise moment after infection with influenza. “This allowed us to ask what HIF-1α is actually doing in the lung, rather than during the initial immune response elsewhere in the body,” explains Jean de Lima, first author of the study. When the team turned off HIF-1α in these T cells, the local immune network collapsed. The T cells produced less of the signaling molecule IL-21, causing the presence of macrophages, other T cells, natural killer cells, and antibody-producing B cells in the lung to decline. This left the lungs poorly equipped to fight off a second infection with a different influenza strain, a challenge that a healthy immune memory would normally handle with ease.
Interestingly, this coordinated response appears to extend beyond viral infection. The researchers also found these HIF-1α driven T cells in a mouse model of lung cancer. In this context, these cells supported the immune system’s fight against tumor cells, suggesting a broader role in protecting the host.
A step toward inhalable vaccines
The study offers a clearer picture of the role these miniature immune hubs play in the lung. Such structures form in tissues throughout the body during infection, cancer and chronic inflammation, but their function has been something of a mystery. These findings suggest they are more than just local antibody factories – they are command centers for broad and coordinated immune protection.
This insight could guide the design of inhalable vaccines to build immune defense at the site of virus entry and open new perspectives for tissue targeted immunotherapies.
Original publication
Jean de Lima et al.
HIF-1α⁺ CD4⁺ T cells coordinate a tissue-resident immune cell network in the lung.
Immunity (2026), DOI: 10.1016/j.immuni.2026.01.023